Monoterpenoid indole alkaloids (MIAs) account for a important group of natural products. Both their highly complex chemical structures and pronounced pharmacological activity have made research on alkaloids, including elucidation of their biosynthetic pathways, very attractive for many decades. MIAs have been proposed to arise from strictosidine, which itself originates from the condensation of tryptophan with secologanin in a 1:1 ratio.
In the phytochemical research on Alstonia rostrata, Dr. CAI Xianghai and Prof. LUO Xiaodong and others from KIB revealed a potential new precursor for these compounds. The alstrostines A and B was isolated and it was determined that they were derived from tryptophan and secologanin in a 1:2 ratio, which supported the presence of a new type of MIA precursor (Organic Letters 2011, 3568). Besides, 6/7-seco rearranged spiro-indolone alkaloids, meloyunines A and B and a monoterpenoid quinoline alkaloid meloyunine C together with its possible intermediate 14,15-dehydromelohenine B, and their precursor 14-vincamenine were isolated from Melodinus yunnanensis. To the knowledge, this is the first report of the co-occurrence of monoterpenoid indoles, a quinoline, and their key intermediates in same plant, supporting the biosynthesis of quinoline from indole and concurring with previous in vivo tracer experiments in the literature (Natural Products and Bioprospecting 2011, 1, DOI 10.1007/s13659-011-0001-0).
In addition, LUO Xiaodong’s research group recently has made a series of progresses in structural diversity and bioactivities, and the results were published on Organic Letters (2010, 968-971; 2009, 4834-4837;2008, 577-580;2007, 1817-1820), Journal of Natural Products (2011, 1161-1168;2010, 22-26; 1075-1079;2009, 1836-1841),Journal of Ethnopharmacology (2010, 174-181; 293-298),Planta Medica (2009, 1537-1541), Helvetica Chimica Acta (2011, 178-183;2010, 2037-2044), Zeitschrift für Naturforschung B (2010, 1164-1168). Promising antitumor indole alkaloids (in Vivo) were also applied for their patents (PCT/CN2010/001775, 200910095137.9;201010104016.9,201010101733.6) .
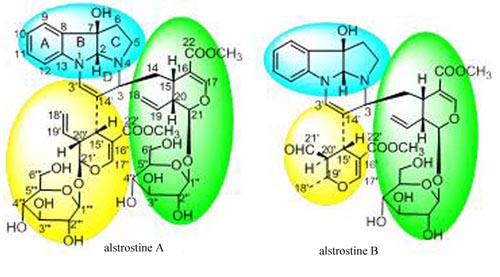
The Structure of alstrostines A and B (Image by LUO Xiaodong CAI Xianghai)
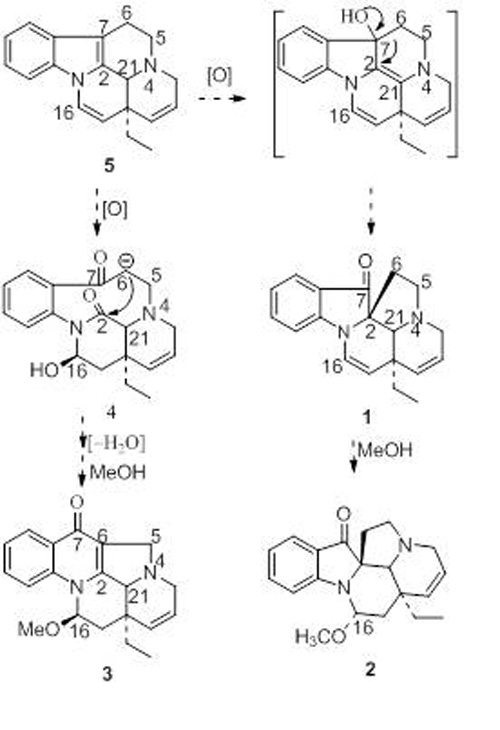
Proposed biogenetic relationships of compound1-5 isolated from Melodinus yunnanensis (Image by LUO Xiaodong CAI Xianghai)




